Published on 16 October 2021
It all began over a meal. When Prof Lawrence Ho, Senior Consultant, Division of Gastroenterology and Hepatology, Department of Medicine, National University Hospital, and Director, Centre of Innovation for Healthcare, National University Health System, and his colleague received funding for a project, they decided they should mull over pertinent medical issues the best way they knew how - over dinner.
Ironically, amidst the Thai fare, the team, including Prof Louis Phee, National Technological University (NTU), Dean of Engineering, concluded that they would focus their efforts on a weight loss innovation.
“The funding agency had asked us to improve on the diagnostic capsule, however, we thought we should explore further possibilities. I thought about doing something for obesity as we know that a lot of people face weight problems,” said Prof Ho.

The birth of an idea
Referencing the fundamental workings of the intragastric balloon procedure, a current weight management practice that endoscopically inserts a balloon into the stomach of a patient, filling up the space and influencing satiety in the body, the EndoPil similarly adopts the balloon-in-stomach concept, with a twist.
Prof Ho and Prof Phee pondered upon the engineering possibilities of refining the process through the ingestion of a balloon. He shared: “The idea came about by thinking about floating up a person with a balloon. It was a joke, but my engineering colleague thought it was a good idea - what if we could swallow the capsule and then have it blown up like a balloon?”
The big challenge was how to swallow the capsule, inflate it, and then have it expelled without a catheter and an endoscope. This critical uncertainty meant that they could avoid undergoing surgery, and also allow for the administration to be as simple as swallowing a pill.
“The engineering challenge was not to pump the balloon in from the outside. There is literally no external help in how we put the capsule in and how we take it out, which is the big difference between the EndoPil and the current intragastric balloons.” said Prof Ho.
Groundbreaking innovations
That momentous meal took place in 2009. Having proven that the ingestion method was plausible in a human volunteer in 2019, and that it was possible to magnetically inflate the balloon through a chemical reaction, the next phase was the “game-stopper phase, to see if we could translate it from a research prototype to a commercial product,” Prof Ho shared.
Two years on, the team has taken big steps to move from the concept stage to a real-life solution. There was one critical step that would make or break the EndoPil - “the manufacturability of its three critical components,” said Prof Ho.
Determining the manufacturability meant figuring out the exact proportion of gases to be contained within the balloon, and if they could be consistently manufactured in the same way.
“The human body can only tolerate [ingesting] a pill of a certain size, beyond which you cannot swallow. So, we had to squeeze in as much substance into the smallest sized pill possible. Once it is in the stomach, it has to be blown up to the maximum volume of gas. We eventually settled on a consistent proportion of substances,” revealed Prof Ho.
With approximately 20% of patients reporting nauseating side-effects from the current intragastric balloon procedure, the team designed the EndoPil to be ingested progressively over a duration, regulating the amount of gas each time so as to reduce the extent of the inflation.
“The current intragastric balloon procedure pumps 500ml of liquid into the balloon within ten minutes. There’s bound to be an intolerance because of the sudden, massive inflation. It’s similar to a car’s airbag exploding. The EndoPil is different because we’ll only pump in up to 120ml, after which we’ll pump in more the next day, allowing for a gradual increase,” he explained.
Their next challenge was to find a material for the capsule that would not only be suited for ingestion, but also one that could withstand stomach acids and the pressure of being inflated.
“We had to make sure that the capsule is bio-compatible, meaning that it would need to be non-toxic to the patient and also able to withstand stomach acidity. Secondly, it has to withstand the pressure of the expansion. This material needs to be impermeable to gas, otherwise it will leak after a month,” explained Prof Ho.
Lastly, the team had to develop a viable activation mechanism, ensuring that the magnet will activate and deactivate the balloon accurately and consistently when located near the capsule.
How it works?
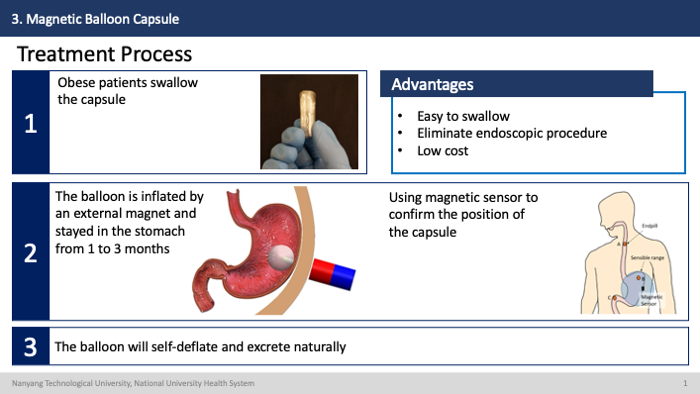
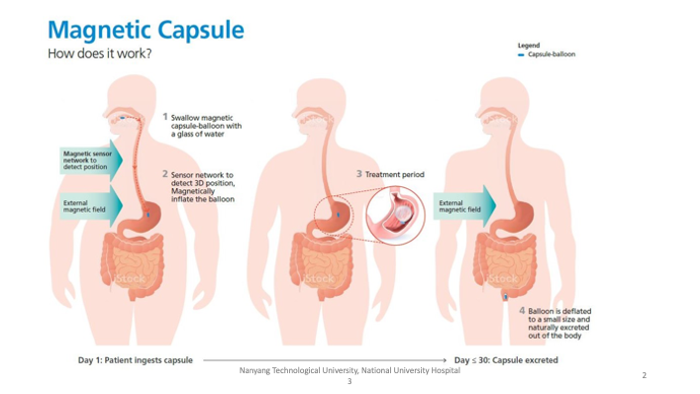
The efficacy test
Prof Ho speculated that the EndoPil could help to reduce body weight by approximately 10%. To ensure that the stomach doesn’t normalise the balloon’s presence over time and reduce its effect, the EndoPil is expected to be expelled after about one month.
“Our body’s natural response is to fight starvation. When you start to lose weight, the body senses that you’re starving. Since human survival depends on you eating, there’s almost no evolutionary mechanism to fight eating the opposite way, hence people gain weight easily,” he pointed out.
As such, the EndoPil will also be prescribed alongside long-term weight management support, such as an accompanying dietary plan, in order to achieve sustained weight loss results.
Next steps
While the EndoPil’s true efficacy is yet to be proven, the capsule’s success would be a revolutionary step forward for individuals with obesity problems.
The team still has to go through many more stress tests to ensure the product works as a whole. “The next phase is to come up with manufacturable prototypes, to be further tested in an integrated fashion. We had three components tested individually so now it’s time to put all the elements together before we can actually test it out on people.”
As for running human trials, Prof Ho emphasised on how pertinent the next experimentation chapter will be. “It will be about two more years before we attempt human trials. The next phase is the safety phase, which is the most important one of all,” he said affirmatively.
In consultation with Prof Lawrence Ho, Senior Consultant, Division of Gastroenterology and Hepatology, Department of Medicine, National University Hospital and Director, Centre for Innovation in Healthcare, National University Health System, Singapore.